The behavior of factor IX in the body is complex
Factors VIII and IX follow different patterns of movement in the body1-4
-
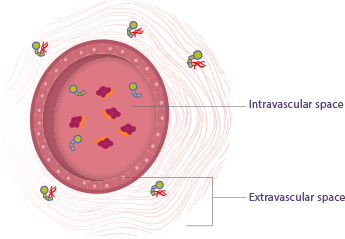
Endogenous factor IX distributes outside of the plasma and
into areas such as the tissues, muscles, and joints, which is also called the extravascular space -
Factor VIII, however, is largely limited to the plasma,
also called the intravascular space
-

Factor IX binds to type IV collagen,
which is located outside of the plasma5 -

Factor VIII binds to von Willebrand,
a protein located inside the plasma2-4
-

Factor IX binds to type IV collagen,
which is located outside of the plasma5 -

Factor VIII binds to von Willebrand,
a protein located inside the plasma2-4
Type IV collagen binding may explain the activity of factor IX in the extravascular space and play a role in coagulation, as shown in preclinical data6
- Additional research is needed to confirm these findings
This distinct behavior means that a single PK parameter alone may not reflect all of the factor IX activity in the body7,8
Preclinical data are not correlated with outcomes. Animal studies may not apply to humans. Additional research for preclinical data and research in humans is needed to confirm these findings.
PK=pharmacokinetic.
 WATCH NOW:
WATCH NOW: 
